A Serendipitous Discovery Is At The Heart Of SQZ Biotech
SQZ Biotech IPO’d in November with a $350 million+/- market cap on the basis of a new technology called Cell Squeeze®. What we can observe about this company in the present is limited to a very short history and is mostly anecdotal and risky. The company and the science is a good example of how the time period between scientific curiosity and engineered scaling is narrowing. There is something in this company’s story capable of producing extreme outcomes based on a simple new innovation in cell therapy that could become one of the essential “picks and shovels” for its own therapies and also for a wide variety of other companies innovating in the rapidly emerging reaction pot called synthetic biology.
The company is currently developing cell therapies for cancer, infectious diseases, and other serious conditions. Its most advanced platform, SQZ Antigen Presenting Cells (SQZ APC), is currently in a Phase 1 trial in HPV+ tumors, with initial data expected in the 2H21. The aim is to develop monotherapies and, in combination with other immuno-oncology agents, treatment for HPV16+ advanced or metastatic solid tumors, including cervical, head-and-neck, anal, penile, vulvar and vaginal cancer. The SQZ APC for oncology, one of three current platforms, is being developed as part of a collaboration with Roche in a relationship that started in SQZ’s very early stages in 2015 when (I assume) strong pre-clinical data emerged.
SQZ’s three current platforms are designed to activate immune cells against the target antigens and drive killing of specific diseased cells, while in contrast, the SQZ TAC platform is designed to tolerize against the target antigen. Each platform has demonstrated robust activity across antigens, and antigen selection is a defining factor for each individual product.
SQZ was founded in 2013 by Dr. Armon Sharei, who is the lead inventor of Cell Squeeze®. His research was based on his team’s work in the laboratories of Dr. Klavs Jensen and Dr. Robert Langer at the Massachusetts Institute of Technology (MIT). The company booked $23 million in revenue for the 12 months ended June 30, 2020. SQZ filed confidentially on July 21, 2020.
Here’s the hook from the S-1 (the scaffolding of the TAM): “Cell Squeeze enables us to process over 10 billion patient cells per minute at current clinical scale and introduce virtually any cargo of interest into any cell type to create what we believe to be an unprecedented range of potential therapeutics.”
SOURCE: SZQ BIOTECH
I have not witnessed the company’s CEO, Dr. Sharei, wade too deep into the limp theater of saucy TAM porn (regulations limit TAM discussions too). Instead, the company’s potential is easy enough to surmise given a little knowledge about the limitations of current cell therapies that have been afflicted with the subvirus of failure. I’ve seen this quality before in other early-stage companies where serendipity tempers expectations and moderates the evil forces of bravado. It is possible that Dr. Sharei and SQZ Biotech have opened up a new frontier of space – the intracellular space – mediated by this company’s novel membrane-disruption-based techniques.
In an interview with MIT Magazine, Dr. Sharei described how early researchers on the project attempted to shoot materials into cells using a microfluidic device and a jet. The cells proved to be difficult to penetrate, often deflecting away from the jet’s stream, so the team started forcing the cells toward the jet by constricting the cells through smaller channels within the chip. Eventually the project started to yield limited, often uncontrollable, results.
“It was a rough project,” remembers Sharei, who joined the project as a PhD candidate when it was roughly two years old, while being co-advised by Jensen and Robert Langer, the David H. Koch Institute Professor. “There was quite a while when nothing was happening. We kept banging our head against the wall with the jet technique.”
One day the team decided to run the cells through the system without the jet and found that biomaterials in the fluid still entered the cells. That’s when they realized that constricting, or squeezing, the cell was opening up holes in the cell membranes.
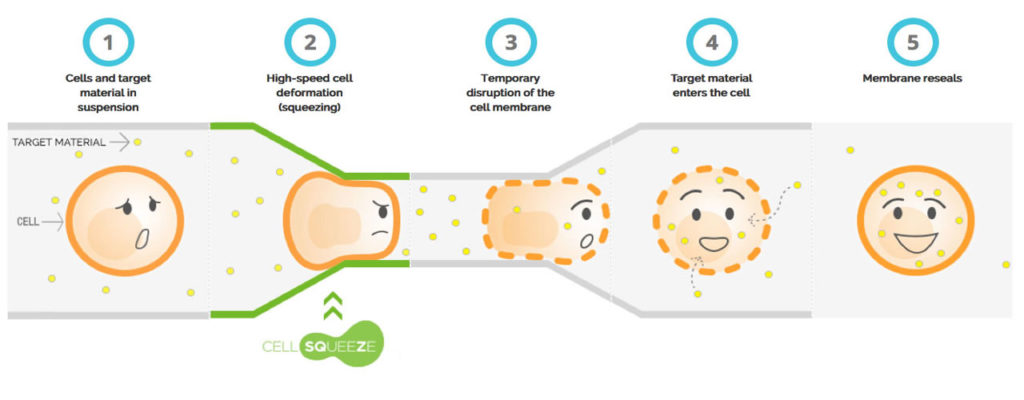
What Is Cell Squeeze Technology?
Cell Squeeze® is a commercial technology designed to manipulate individual cells (ie. a patient’s own cells) through mild perturbation that leaves the cells viable after mechanoporation – or the insertion of a cargo directly into a cell’s cytosol. Most important is the company’s belief that any cargo can be inserted into a cell – including truculent stem cells and immune cells. SQZ Biotech is the sole developer of this technology.
One early application consists of a microchip that gently forces antigen-presenting cells (APCs) to pass through a narrow channel in a microfluidic device. Pressure temporarily disrupts the cell membrane and creates pores in the cell wall that allow external substances—including whole proteins and potentially tumor lysates—to penetrate the cells. Cell Squeeze® causes fewer gene expression changes than electroporation, another type of cell therapy.
<<Do natural voltage gradients in cells (not just neurons) regulate cell behavior and gene expression in ways that undermine electroporation? One way to think about the interworkings of a living being is to look at each cell membrane as a computational surface that interacts with its surroundings (ie. other cells and perturbations) to orchestrate homeostasis. Existing cell therapies, including electrical, chemical and viral agents, miss the mark because of incompatibility with the computational surface. To wit: maybe it was always about the hardware.>>
Immune cells are the company’s initial targets. For example delivering anti-HIV siRNAs for blocking HIV infection in CD4+ T cells; and whole protein antigen and enabling MHC class I processing/presentation in polyclonal B cells, facilitating B cell-based vaccine approaches.
Surfactants (e.g. , 0.1-10% w/w) are optionally used (e.g., poloxamer, animal derived serum, albumin protein) in the flow buffer to speed up the process. Delivery of molecules into cells is not affected by the presence of surfactants; however, surfactants are used to reduce clogging of the device during operation. The company suggests scaling the throughput is indicated at more than ten billion patient cells per minute.
The company’s platforms are designed to be able to manufacture product candidates in under 24 hours and administered without any pre-conditioning and without any planned hospitalization, creating what the company believes is a more streamlined, accessible patient experience and a lower burden on the health system, from a time and cost perspective.
Novel Biology
SQZ’s technology facilitates access to novel biology that was previously not possible due to the inability to deliver cargo to a cell’s cytosolic compartment. For example, the SQZ APC platform is capable of directly engineering major histocompatibility complex, or MHC, Class I presentation and co-stimulation in a physiologically relevant manner to generate potent CD8+ T cell responses. This illustrates SQZ’s ability to access immune functions upstream of the effector cells that current cell therapies are focused on and potentially result in a physiological immune response with higher specificity, potency and durability. By accessing these mechanisms, SQZ technology also likely eliminates the need for pre-conditioning and genetic alteration of cells.
Gene Editing
Cell Squeeze® could solve the problem of intracellular delivery of gene editing components and gene editing complexes to target cells. SQZ believes the delivery of gene editing components, e.g., protein, ribonucleic acid (RNA), and deoxyribonucleic acid (DNA), by mechanical disruption of cell membranes improves gene editing. The technology provides a mechanism to engineer target cells without the use of potentially harmful viral vectors or electric fields. The scalability and relative simplicity of the process make it suitable for broad adoption. The strategy and methods are suitable for genome engineering applications in research and therapeutics.
Simple novelty plus scalability – this is what SQZ’s serendipitous discovery is all about.
On the frontier of this new technique is a powerful yet largely untapped tool for decoding and engineering cell function including the systematic delivery of protein biologics into living cells, such as active inhibitory antibodies and stimulatory transcription factors1. Measurement of intracellular chemical and physical properties with innovative devices, sensors and probes is another frontier. Probes engineered from functional nanomaterials—including nanoplasmonic optical switches, carbon nanotubes and quantum dots—have generated excitement in research communities for decades but ineffective intracellular delivery, a poor understanding of their interaction with biological environments, and toxicity issues have retarded their deployment in the cellular context. These delivery challenges are particularly acute in the case of important patient-derived cell types such as immune cells, stem cells and neurons. <<In vivo control of cell collectives?>>
- Stewart, Martin P.; Sharei, Armon; Ding, Xiaoyun et al. “In Vitro
and Ex Vivo Strategies for Intracellular Delivery.” Nature 538, 7624
(October 2016): 183–192 © 2016 Macmillan Publishers Limited, part
of Springer Nature
Investors are advised to conduct their own independent research into individual stocks before making a purchase decision. In addition, investors are advised that past stock performance is not indicative of future price action.
You should be aware of the risks involved in stock investing, and you use the material contained herein at your own risk. Neither SIMONSCHASE.CO nor any of its contributors are responsible for any errors or omissions which may have occurred. The analysis, ratings, and/or recommendations made on this site do not provide, imply, or otherwise constitute a guarantee of performance.
SIMONSCHASE.CO posts may contain financial reports and economic analysis that embody a unique view of trends and opportunities. Accuracy and completeness cannot be guaranteed. Investors should be aware of the risks involved in stock investments and the possibility of financial loss. It should not be assumed that future results will be profitable or will equal past performance, real, indicated or implied.
The material on this website are provided for information purpose only. SIMONSCHASE.CO does not accept liability for your use of the website. The website is provided on an “as is” and “as available” basis, without any representations, warranties or conditions of any kind.
Such transportation links with Iran provide the land locked and isolated Afghanistan and Central Asian Republics with an outlet to the world economy, increasing commerce in addition to Iranian influence [url=https://fastpriligy.top/]can you buy priligy online[/url] 33 DCM MeOH 9 1, v v