A Game-Changer in ED Therapy: Rapid-Onset Nasal Spray
LTR Pharma Ltd (LTR) is an Australian biotech company developing in a novel nasal spray to treat erectile dysfunction (ED).
I interviewed the company’s CEO – see below.
Lead Indication
LTR’s lead indication is the treatment of ED through its proprietary product, SPONTAN®. SPONTAN® offers a novel delivery method, bypassing the gastrointestinal tract and enabling rapid absorption through the nasal mucosa. The nasal spray uses vardenafil, a phosphodiesterase type 5 (PDE5) inhibitor, which is already approved for oral use in treating ED under brand names Levitra® and Staxyn®. PDE5 inhibitors are a group of medications used to treat physiological causes of ED and today are the standard of care. Viagra® and Cialis® are other PDE5s inhibitors in pill form. Vardenafil is a Bayer drug and is off-patent.
LTR’s new therapy will be a “first-in-class” rapid onset, on-demand therapeutic nasal spray for the treatment of ED.
Problem/Solution
Problems with existing oral administration PDE5s include: do not work for one third of patients (sometimes attributed to taking pill after eating), long response time, adverse reactions for one third of patients leading to high discontinuation rate.
Advantages/solutions offered by SPONTAN®: rapid onset of action (10 minutes or less), higher absorption rate means lower active ingredients needed (and less adverse reactions), less degradation due to bypassing of digestive system.

Drug Development History
LTR has in-licensed on a global exclusive basis all rights to develop, manufacture, commercialize and sell SPONTAN® as a nasal spray product. LTR collaborated with founding researcher from California, Professor Moses Chow, who developed the proprietary compound SDS089, now called SPONTAN®. Professor Chow was awarded his Doctor of Pharmacy in 1972 from the University of California, San Francisco, USA; and he completed his residency in Clinical Pharmacy at VA Hospital, Palo Alto, USA.
Currently, Professor Chow is Professor of Pharmacy Practice and Director of the Center for Advancement of Drug Research and Evaluation (CADRE), College of Pharmacy at the Western University of Health Sciences, Pomona, USA. He is also an adjunct professor at the Chinese University of Hong Kong, China.
Professor Eric Chung, LTR’s scientific advisor, was awarded The Emil Tanagho Prize in 2023 at the International Society for Sexual Medicine (ISSM) and the Middle East Society for Sexual Medicine (MESSM). His winning research was titled, “Can Novel SDS-089 Nasal Vardenafil Spray Solution Achieve Satisfactory Drug Plasma Level Similar to Oral Vardenafil Formulation? A Bioanalysis Study Comparing Vardenafil Nasal vs Oral Formulations Using Liquid Chromatography Tandem Mass Spectrometry.”
Cash, Catalysts & Balance Sheet
LTR had A$5.3 million on hand as of March 31, 2023 and is burning about A$1 million per quarter now that its Australian bioequivolency study is complete.
Shares outstanding: 139 million
Market cap as of 7/4/24: A$108 million (US$73 million), no debt
LTR’s IPO in December 2023 (ASX:LPT) raised A$7 million. LTR was spun out of LTR Medical to develop SPONTAN®.
Contingent Liabilities
On June 30th 2023, the company reported contingent liabilities which exist in relation to potential milestone payments arising under its Strategic Drug Solutions Inc (SDS) license agreement for the SPONTAN® assets. As of the company’s IPO, contingent liabilities totaled US$4.0 million and are dependent upon the granting/approval of relevant patents which are the subject matter of the SDS license agreement, as well as future decisions regarding the clinical focus of the company which may depend on the success of the company’s clinical research/trials.
Subsequent to June 30th, 2023, LTR agreed to pay US$500,000 and a further amount of US$300,000 under a variation to the SDS license agreement. The aggregate of the remaining milestone payments which may become payable under this agreement subject to the achievement of certain milestones is US$3,000,000, net of the loan of US$200,000 which the company previously paid under the license agreement. As of March 31, 2024, LTR records A$475,097 unpaid to SDS under its IPO allocation “intention.”
The top three shareholders are:
- LTR Medical Pty Ltd: 33%
- Lee Rodne (CEO): 4.5%
- Strategic Drug Solutions, Inc: 4.25%
Here are the catalysts that are likely to occur in the next 12 months:
- initiation of sales in Australia under TGA’s special access schemes (SAS) or Authorized Prescriber Scheme (APS), prior to full approval – likely within months;
- USA stock listing, modest capital raise and initiation of coverage by a USA sell-side analyst, all of which are likely to occur concurrently;
- partnering/licensing agreement with large pharma company;
- commencing of FDA 505(b)(2) expedited route to market under the change of route to administration for an already approved drug;
Commercialization & TAM
The company is seeking to use the shortened Therapeutic Goods Administration (TGA) (Australia) Type F and similar FDA (USA) 505(b)(2) pathways that govern commercialization under the change of route to administration for an already approved drug, or bioequivalency, which largely de-risks the pharmacokinetics (PK) dimension of approval. The company’s pivotal Australian bioequivalency trial is complete and showed positive results, as expected.
LTR plans to start selling the drug in Australia under TGA’s special access schemes (SAS) or Authorized Prescriber Scheme (APS) in the coming months, prior to full approval. This special path allows medical practitioners to become authorized to prescribe a specified unapproved therapeutic good to a group (or class) of patients with a particular medical condition. LTR expects full drug approvals in 2025.
Once approved in the USA and Australia, the company plans to go to market through the traditional route of physician prescriptions and through men’s health websites, such as Ro.co, that market directly to consumers via telemedicine. The telemedicine channel allows rapid uptake of new patients and lowers access barriers to ED treatment.
LTR’s nasal spray product is changing the route of administration of vardenafil from an oral delivery to an intranasal delivery, offering a significant opportunity given:
- large existing market opportunity for ED medications;
- growth in prevalence of ED, primarily caused by growth in aged cohorts, with ED prevalence increasing with age and poor metabolic health;
- known side effects of existing pill products;
- increased concern of ingesting compounds, with lack of therapeutic benefits for a large cohort of patients; and
- decreased regulatory burden and costs through the anticipated 505(b)(2) submission pathway for the FDA, and Type F application for the TGA. Either of these regulatory agencies may request LTR to complete animal studies to test other characteristics of SPONTAN®, such as the toxicology of vardenafil as an intranasally delivered drug, which may delay its approval. Animal studies of this nature are typically complete within a few months.
LTR will seek to enter into sub-licensing arrangements with third parties allowing them to manufacture and/or sell SPONTAN® in any jurisdiction in which the company does not intend to directly operate in.
Pricing
The company is targeting between 16-18 doses per bottle at a cost of AU$100-125 per bottle which is similar to oral PDE5 inhibitor prices.
TAM
The global market for ED is substantial and growing. In 2021, the market was valued at approximately $3.6 billion and is expected to reach $5.9 billion by 2028. The demand for ED treatments is driven by an aging population, increasing prevalence of risk factors such as diabetes and cardiovascular diseases, and growing awareness of sexual health issues. Global estimations of the prevalence of ED vary widely, depending on the study population and definition of ED used. Population-based studies have variously reported prevalence ranging from as low as 3.0% to as high as 76.5%. A study across eight countries amongst males aged 40-70 indicated prevalence ranging from 42.1% in Brazil to 52.2% in Italy, with a total self-reported prevalence of 40.5% across all age cohorts. (Estimates cited by a report produced by Frost & Sullivan).
The Massachusetts Male Aging Study (MMAS) reported that about 52% of men between 40 and 70 years old experience some form of ED. The British Journal of Urology International published a study in 2000 estimating that by 2025, around 322 million men worldwide will suffer from ED.
Potential Other Indications and Dosages
LTR intends to develop a range of new nasal spray products both for the treatment of ED and new indications by investing in product R&D. This includes different concentrations/dosages of SPONTAN® to address market needs and additional nasal spray products using other approved PDE5 inhibitors to create a range of nasal spray products for the treatment of ED and non-ED conditions.
Clinical Progression
In February 2020, LTR completed an investigator lead, human proof of concept study titled “A Randomized, Single-Dose, Cross-over, Bioavailability Study to Evaluate SDS-089 Solution as Nasal Spray in Comparison to Levitra Oral Tablet 10 mg in Healthy Volunteers” for SDS-089, or SPONTAN®. This study, completed by Western University of Health Sciences in California, was designed to compare the plasma concentration levels of vardenafil in healthy male subjects.
The study was a randomized, single dose cross-over study of males aged between 24 to 45 with 12 healthy participants. The delivery of the SDS-089 nasal spray solution used a 100 ul per dose nasal spray device manufactured by AptarGroup Inc. The study assessed the plasma concentration levels of vardenafil HC1 in healthy male subjects comparing Vardenafil® HC1 as SDS-089 nasal spray (4 mg) and an oral vardenafil tablet (10 mg).
The study was published in May 2023 in The Journal of Sexual Medicine and confirmed the rapid onset of effect for SDS-089 is approximately 10 to 15 minutes compared to up to 60 minutes which is typical of existing oral ED drugs. Based on the pharmacokinetic data, this study showed positive characteristics / attributes for SDS-089, including:
- SDS-089 showed faster Tmax (the amount of time a drug is present at maximum concentration in a patient’s serum) suggesting rapid onset via nasal spray delivery (≤ 10 minutes);
- SDS-089 shows a Cmax (the peak concentration of a drug in a patient’s serum) within 10 minutes suggesting patient will respond shortly after administration;
- no severe adverse events being detected;
- and an acceptable safety profile for SDS-089.
Bioequivolance Trial
The company recently conducted a pivotal trial in Australia: “A Phase 1, Randomized, Open-label, Single-dose, Two-period, Two- treatment, Cross-over Study Comparing the Pharmacokinetics of Vardenafil Following Administration of SDS-089 Nasal Spray and Levitra Tablet in Healthy Male Adult Subjects.” The strength of this study is its randomized crossover design. It was a single-dose, randomized, open-label 2-treatment, 2-period crossover study of SPONTAN nasal spray (5 mg Vardenafil: a single 2.5 mg spray in each nostril compared to Vardenafil tablets 10 mg.
In June 2024, LTR reported interim results of this trial:
- Initial results show SPONTAN® achieves rapid absorption and faster onset of action compared to oral PDE5 inhibitors (i.e., vardenafil, sildenafil, tadalafil);
- SPONTAN’s nasal spray technology delivered a similar amount of drug (Cmax) at half the dose of the oral PDE5 and was significantly faster;
- Better safety profile for SPONTAN® compared to oral PDE5 inhibitor dosing;
These positive data results will be used for regulatory filings in key markets.
LTR has validated the US FDA’s 505(b)(2) regulatory pathway through an expert regulatory review.
Treatments for ED
Existing first-line treatments primarily include oral PDE5 inhibitors as the gold standard pharmacological products, and lifestyle modifications. However, a significant portion of patients with ED are non-responsive to PDE5 inhibitors or are not able to take oral medications. Many patients abandon PDE5 treatments due to the length of time of response (often one hour) or because of non-effectiveness due to taking the drug with a full stomach.
Existing second- and third-line treatments include injectable vasodilator agents, vacuum constriction devices (VCDs), intracavernosal injections (ICIs), intraurethral suppository of prostaglandin E1, and penile prosthesis implantation (PPI). Additionally, a topical gel (MED3000, marketed as Eroxon® Stimgel) was commercially launched in the EU in 2022 following approval as a medical device, and US approval is currently ongoing.
Management Team
LTR has an experienced management team with a track record in pharmaceutical development and commercialization. The team is led by Executive Chairman Lee Rodne, who brings extensive experience in healthcare management and commercialization. Non-Executive Director Dr. Julian Chick has a background in life sciences and corporate development, providing strategic oversight and expertise. Non-Executive Director Maja McGuire adds legal and governance acumen to the board.
The company also benefits from a robust Scientific Advisory Board, composed of experts in pharmacology, clinical research, and regulatory affairs.
Intellectual Property
LTR’s intellectual property portfolio is centered around the unique formulation and delivery method of SPONTAN®. The company has filed patent applications in key markets to protect its proprietary technology and ensure competitive advantage. These patents cover various aspects of the nasal spray formulation and its use in treating erectile dysfunction.
Here are the patent filings, according to LTR:
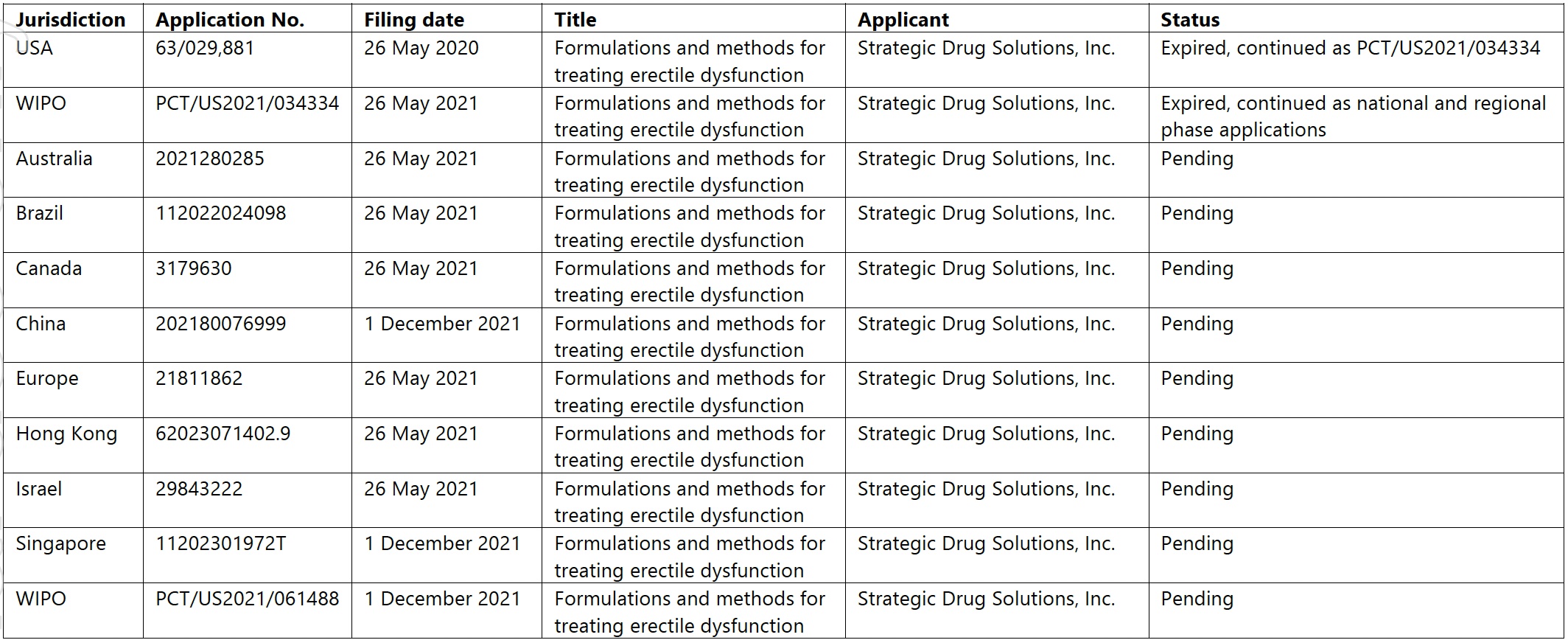
Source: LTR Pharma
The company holds a licensing agreement with SDS, the developer of the intranasal delivery technology used in SPONTAN®. This agreement grants LTR exclusive rights to commercialize the technology for the treatment of ED. The term of the SDS license agreement commenced on October 1, 2020. The term expires on the later of September 30th, 2040 or the date all patents issued under the patent rights have expired and the patent applications under the patent rights (if any) are cancelled, withdrawn or abandoned.
Pfizer’s initial attempts to develop a nasal formulation of sildenafil, the active ingredient in Viagra, were marked by the filing of patent EP0967214B1 in 1999. This patent covered a pharmaceutical composition suitable for nasal administration aimed at providing a rapid onset of action and improved bioavailability compared to oral formulations. Pfizer never successfully brought a nasal sildenafil product to market, even when faced with Viagra’s (sildenafil) patent expiration. The reasons behind this include significant technical challenges such as maintaining the stability of sildenafil in a nasal formulation and ensuring effective absorption without causing irritation or damage to the nasal lining.
LTR’s compound succeeded in developing a viable nasal spray for erectile dysfunction. They focused on vardenafil which offered better performance characteristics and stability for nasal administration. The research group Strategic Drug Solutions (SDS) initially faced similar challenges with sildenafil but pivoted to vardenafil after encountering stability issues. Over several years of research, they perfected the formulation and developed Spontan, a nasal spray that LTR Pharma subsequently acquired the global rights to. This success was due to their ability to maintain stability and prevent crystallization in the solution, which were significant obstacles for Pfizer.
Patent WO 99/66933, which pertains to nasal formulations of sildenafil, serves as another example of intellectual property failing to produce a viable drug. This patent, although promising in its description of nasal administration methods for sildenafil, did not lead to a commercial product due to the same technical difficulties that Pfizer encountered. This patent appears to have expired in 2019.
Manufacturing
LTR has contracted with Mayne Pharma Group Ltd (ASX:MYX). MYX has completed validation steps to meet FDA requirements by conducting stability testing, quality control checks, product purity assessments and packing integrity evaluations. MYX also produced SPONTAN® for its clinical trials in Australia and it expected to produce inventory for the commercial roll-out. MYX is a leading contract manufacturing company in Australia.
Investors are advised to conduct their own independent research into individual stocks before making a purchase decision. In addition, investors are advised that past stock performance is not indicative of future price action.
You should be aware of the risks involved in stock investing, and you use the material contained herein at your own risk. Neither SIMONSCHASE.CO nor any of its contributors are responsible for any errors or omissions which may have occurred. The analysis, ratings, and/or recommendations made on this site do not provide, imply, or otherwise constitute a guarantee of performance.
SIMONSCHASE.CO posts may contain financial reports and economic analysis that embody a unique view of trends and opportunities. Accuracy and completeness cannot be guaranteed. Investors should be aware of the risks involved in stock investments and the possibility of financial loss. It should not be assumed that future results will be profitable or will equal past performance, real, indicated or implied.
The material on this website are provided for information purpose only. SIMONSCHASE.CO does not accept liability for your use of the website. The website is provided on an “as is” and “as available” basis, without any representations, warranties or conditions of any kind.
This is such a useful resource—thanks!
꼼꼼한 먹튀검증을 거쳐 선별된 안전한 메이저 토토사이트 리스트를 확인하세요. 다양한 이벤트와 혜택도 함께 제공됩니다. https://mtverify.com/
You’ve presented the information so clearly, well done!
Your writing encourages open-mindedness and exploration,
Really appreciate the straightforward advice here.
The way you broke this down is incredibly helpful.
I found this post to be extremely helpful, thank you!
I guess most of us haven’t seen a good Recipe blog like my name.
I appreciate how clear and well-written this is.
I really want to let you know about the best 3D Comics site, NewTaker.Com. You should never Check it out if You Don’t Like 3D ER0T!C SE* COMICS.